One hundred years of the Schrödinger equation
1 April 2026
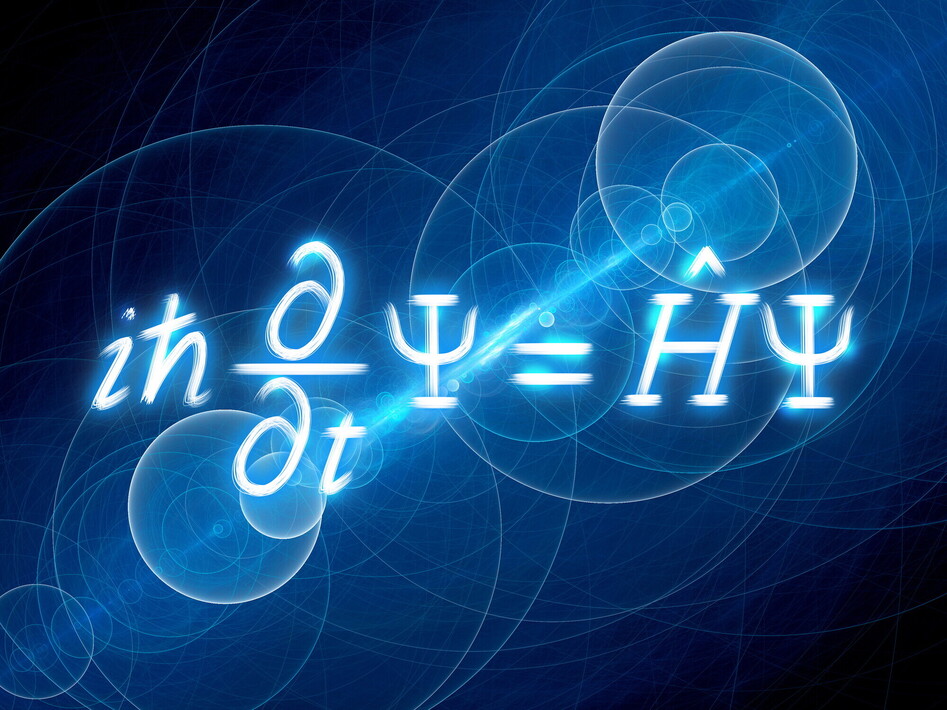
This year marks the centenary of one of the most famous and revolutionary equations in the history of science: the Schrödinger equation. Written in 1926 by the Austrian physicist Erwin Schrödinger, this formula is one of the cornerstones of quantum mechanics, the theory that describes the behaviour of matter and energy on the microscopic scale of atoms and subatomic particles.
But what exactly is the Schrödinger equation? And why, one hundred years on, is it still so important? Put simply, the Schrödinger equation is a mathematical law that describes how the quantum state of a physical system evolves over time. At its heart is a quantity known as the wave function, usually indicated by the Greek letter ψ (psi). This function does not tell us exactly where a particle is, but provides information about the probability of finding it in a certain place or with a certain energy. Here, one of the most surprising ideas in quantum physics emerges: at the microscopic level, nature is not determined in the same way as in Newtonian classical physics. We cannot predict with absolute certainty how an electron will behave, but only calculate the probabilities of its possible behaviours. The Schrödinger equation is precisely the tool that allows us to do this. The importance of the equation lies in the fact that, by solving it for a given system (for example, an electron in an atom), we obtain the so-called quantised energies and the shapes of atomic orbitals. It is thanks to these results that we understand the structure of atoms, the arrangement of electrons and, ultimately, the chemical properties of the elements. Without the Schrödinger equation, we would have no coherent explanation of the periodic table, chemical bonds or how molecules work. Its practical applications are enormous. Quantum mechanics, built in part on this equation, underpins technologies we use every day: semiconductors, lasers, LEDs, magnetic resonance imaging, and even modern computers and smartphones. Today, moreover, the Schrödinger equation is central to research into quantum computing, which promises to revolutionise the way information is processed. Beyond its technological impact, the equation has also had a profound cultural and philosophical significance. It helped overturn the idea that science could describe the world as a perfectly predictable machine, introducing concepts such as probability, indeterminacy and the role of the observer. One hundred years after it was first formulated, the Schrödinger equation remains an extraordinary example of how a single mathematical idea can radically transform our vision of reality. An abstract formula, born on paper, that continues to shape science, technology and the way we think about the invisible world around us.